|
8/31/2023 0 Comments Atomic size trend ap che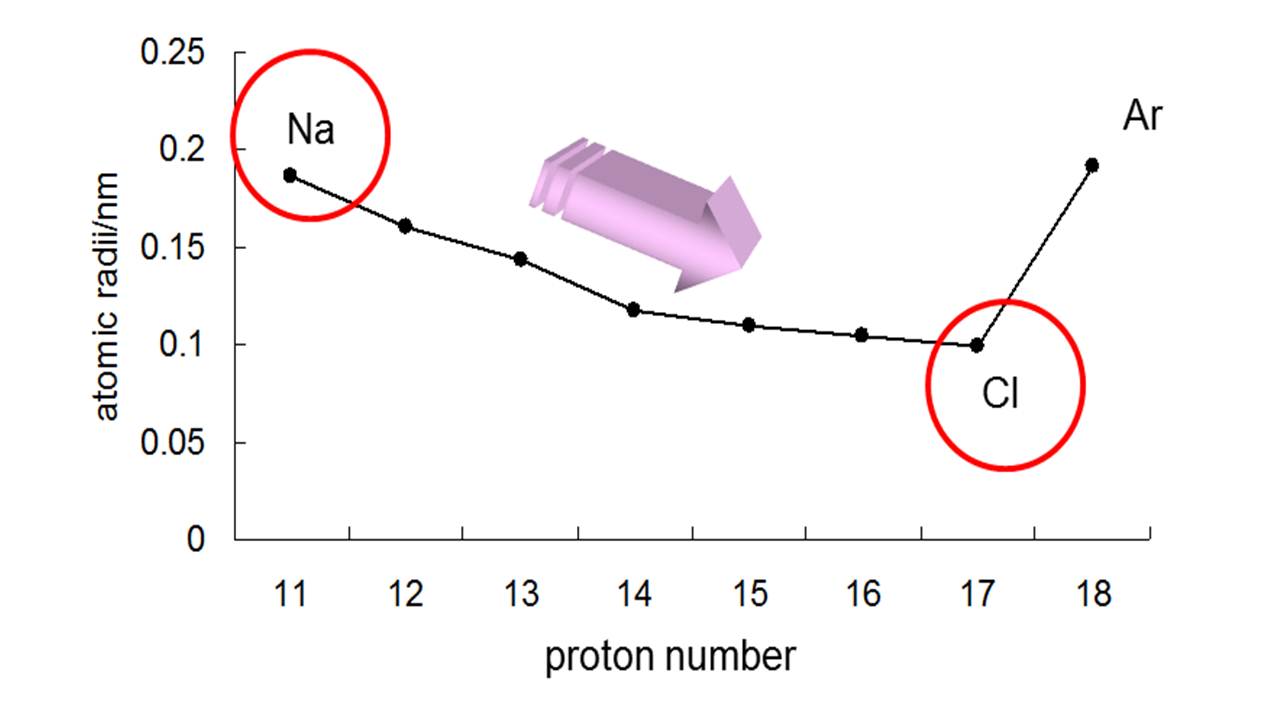
This will cause the electrons push each other away and spread out, causing the atom to become larger. When this occurs there are more electron-electron repulsions and there is a lower net nuclear attraction per electron. Anions are formed when an electron is gained. So, the newly formed ion becomes a more condensed version of its neutral atom. When this occurs there are less electron-electron repulsions and there is a greater net nuclear attraction per electron. C & D: Cations are formed when an electron is lost.However, for less symmetrical and more polar lattices such as those with C n, C nh, and C nv symmetries, significant changes in the electron density can occur, causing deviations from spherical shape these deviations make ionic radii more difficult to measure. For instance, lattices with O h and T d symmetries are considered to have high symmetry thus the electron densities of the component ions occupy relatively-spherical regions and ionic radii can be measured fairly accurately. The point group symmetry of a lattice determines whether or not the ionic radii in that lattice can be accurately measured (Johnson 1973). For a given ion, the ionic radius increases with increasing coordination number and is larger in a high-spin state than in a low-spin state.Īccording to group theory, the idea of ionic radii as a measurement of spherical shapes only applies to ions that form highly-symmetric crystal lattices like Na + and Cl. Ionic radius is not a permanent trait of an ion, but changes depending on coordination number, spin state, and other variables (Shannon 1976). After comparing many compounds, chemist Linus Pauling assign a radius of 140 pm to O 2- and use this as a reference point to determine the sizes of other Ionic Radii (Jensen 2010). However, it is to consistently and accurately determine the proportions of the ionic bonds. The ionic radius of an atom is measured by calculating its spatial proportions in an ionic bond with another ion within a crystal lattice. Measurement and Factors Affecting Ionic Radii This is an example of how these elements behave differently, and a reminder that trends do not necessarily apply to every instance.\( \newcommand\) Two elements decrease and then increase again, seemingly throwing the trend off. On the bottom of the table we see a slight bump in the trend.  
However as we go down the group the situation of the equation is reversed and so our atoms become larger as their core charge gets smaller. This equation fits perfectly with the above diagram and explains why the atoms get smaller as they travel across the period. As this number gets higher, the valence electrons are pulled closer to the nucleus, therefore decreasing the atomic size of the atom. The core charge is simply an expression of the attractive force that the centre of the nucleus gives off to the valence electrons. CORE CHARGE = PROTONS - NON-VALENCE ELECTRONS. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.Ītomic radius can be linked to core charge. With the above image, courtesy of Webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.Ītomic radius is measured from the centre of the nucleus to the outermost electron shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
